NEWS
News Center
Computational power unlocks the mystery of the molecule
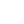
Researchers must study nearly 100,000 analog images of this type to determine what triggered the splitting of water molecules. A lot of computing power has been invested in these simulations.
Chemical reactions always happen around us - in the air we breathe, in the water we drink, in the factories that produce the products we use in our daily lives. These reactions were unexpectedly fast. Under the optimal conditions given, molecules can react with each other in billionths of a second.
The industry has been working hard to achieve faster and better chemical processes. The production of hydrogen, which requires the splitting of water molecules, is an example. To improve this process, researchers need to know how different molecules interact and what triggers these reactions. Computer simulation can study what happens within a billionth of a second, so if you know the order of the chemical reactions, or if the triggers that trigger the reaction happen often, you can study the steps of the reaction.
But the actual situation is often not the case. Molecular reactions often exhibit different behaviors. Optimal conditions often do not exist, such as water molecules used to make hydrogen, which makes it difficult to study the reaction using computer simulations.
Until recently, scientists did not know what triggered the splitting of water molecules. However, it is well known that water molecules have a 10-hour life before splitting. Ten hours may not sound very long, but it is quite long compared to the molecular time scale, one billionth of a second. This makes it extremely challenging to determine the mechanism that causes the water molecules to divide. It's like finding a needle in a haystack.
NTNU researchers have recently discovered a way to identify needles in a haystack. In their research, they combined two technologies that were not used before.
They studied nearly 100,000 analog images of this type before they could determine what triggered the splitting of water molecules. A lot of computing power has been invested in these simulations. Through their special methods, the researchers managed to accurately simulate how water molecules divide. Anders Lervik, a researcher at the NTNU Department of Chemistry, said: "We started looking at the 10,000 analog films and analyzed them manually to try to find out why the water molecules split."
“After spending a lot of time researching these mock movies, we found some interesting relationships, but we also realized that the amount of data was too large to manually investigate everything.” Researchers used machine learning to discover triggering reactions. s reason. This method has never been used for this type of simulation. Through this analysis, they found a small number of variables that describe what triggered these reactions.
Their findings provide detailed knowledge of the causal mechanisms and ideas for improving the process. In this study, an important step has been taken to make industrial chemical reactions happen faster and more efficiently. It offers great potential for increasing hydrogen production.
Contact us
Hotline:
Copyright © 2022 Jiangsu Huada Chemical Group Co., Ltd.
苏ICP备05042815号-1 Website Construction: Chinese Enterprise Power ChangZhou seo

Mobile Website